news
Launch of new reagents: phospho-ubiquitin reagents
The discovery that ubiquitin itself can be modified through phosphorylation by the kinase PINK1 provides a major breakthrough linking the two most important signalling pathways in cells; phosphorylation and ubiquitylation. Parkin and PINK1, the two main proteins associated with Parkinson’s Disease (PD) comprise a mitochondrial quality control pathway that promotes neuronal survival through autophagy of damaged mitochondria in a process known as mitophagy. The accumulation of PINK1 on depolarised or damaged mitochondria leads to the activation and translocation of Parkin to the outer mitochondrial membrane. Phosphorylation of Parkin by PINK1 at Ser65 located in its Ubl domain markedly increases the E3 ligase activity of Parkin resulting in ubiquitylation of proteins on the outer mitochondrial membrane, triggering selective mitophagy.
To enable research into the role and function of phosphorylated ubiquitin proteins, UbiQ used its UbiQ-Syn technology to develop a set of phospho-ubiquitin mutants that have all been identified in cells by proteomics (Figure 1).
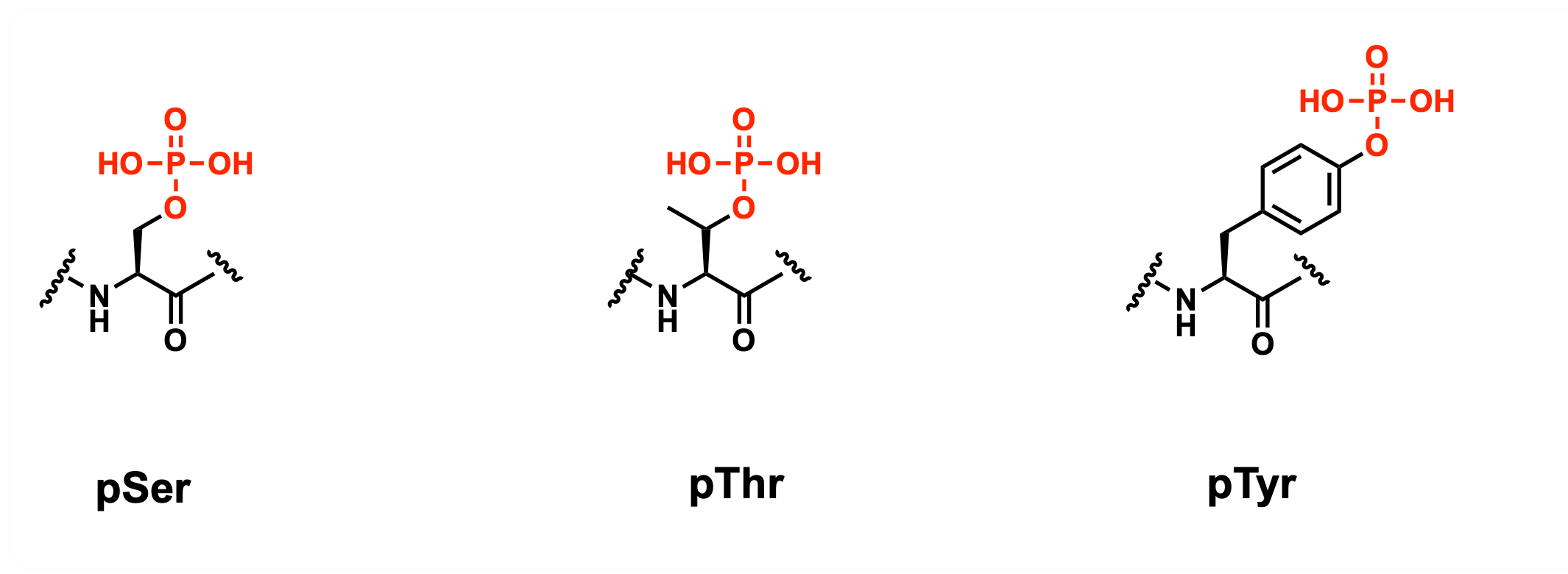
- UbiQ-089: Ub pSer65
- UbiQ-113: Ub pSer57
- UbiQ-091: Biotin-Ahx-Ub pSer65
- UbiQ-092: Biotin-Ahx-Ub pThr7
- UbiQ-093: Biotin-Ahx-Ub pThr12
- UbiQ-094: Biotin-Ahx-Ub pSer57
- UbiQ-095: Biotin-Ahx-Ub pTyr59
Figure 1.
Literature. (1) Kane et al. J Cell Biol 2014, 205, 143. (2) Kazlauskaite et al. Biochem J 2014, 460, 127. (3) Kondapalli et al. Open Biol 2012, 2, 120080. (4) Koyano et al. Nature 2014, 510, 162. (5) V. Sauve and K. Gehring Cell Res 2014, 24, 1025. (6) Spratt et al. Nat Commun 2013, 4, 1983. (7) Trempe et al. Science 2013, 340, 1451. (8) T. Wauer and D. Komander EMBO J 2013, 32, 2099. (9) Yamamoto et al. J Biol Chem 2005, 280, 3390.
