news
Strategy for development of site-specific ubiquitin antibodies
As part of collaborative research between The Netherlands Cancer Institute (NKI-AVL), Leiden University Medical Centre, UbiQ and the University of Amsterdam, van Kruijsbergen et al. report in Frontiers in Chemistry detailed protocols for the development of antibodies recognizing site-specific ubiquitin modifications. Considerations for antigen design (please check out our UbiQ Block technology), screening and quality control are outlined.
Protein ubiquitination is a key post-translational modification regulating a wide range of biological processes. Ubiquitination involves the covalent attachment of the small protein ubiquitin to a lysine of a protein substrate. In addition to its well-established role in protein degradation, protein ubiquitination plays a role in protein-protein interactions, DNA repair, transcriptional regulation, and other cellular functions. Understanding the mechanisms and functional relevance of ubiquitin as a signaling system requires the generation of antibodies or alternative reagents that specifically detect ubiquitin in a site-specific manner. However, in contrast to other post-translational modifications such as acetylation, phosphorylation, and methylation, the size of ubiquitin (76 amino acids) complicates the preparation of suitable antigens and the generation antibodies detecting such site-specific modifications. As a result, the field of ubiquitin research has limited access to specific antibodies. This severely hampers progress in understanding the regulation and function of site-specific ubiquitination in many areas of biology, specifically in epigenetics and cancer. Therefore, there is a high demand for antibodies recognizing site-specific ubiquitin modifications. Here we describe a strategy for the development of site-specific ubiquitin antibodies. Based on a recently developed antibody against site-specific ubiquitination of histone H2B, we provide detailed protocols for chemical synthesis methods for antigen preparation and discuss considerations for screening and quality control experiments.
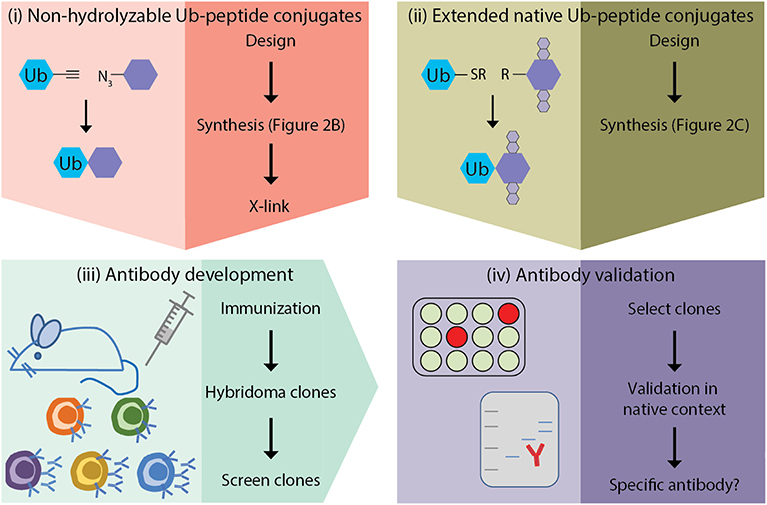
Figure 1. Outline of site-specific Ub antibody development. (i) design and synthesis of non-hydrolyzable Ub-peptide conjugates for immunization; (ii) design and synthesis of extended native iso-peptide linked Ub-peptide conjugates for screening; (iii) immunization, and generation and selection of hybridomas; (iv) selection of clones and antibody validation.
Reference: Ila van Kruijsbergen, Monique P. C. Mulder, Michael Uckelmann, Tibor van Welsem, John de Widt, Aldo Spanjaard, Heinz Jacobs, Farid El Oualid, Huib Ovaa,* and Fred van Leeuwen.* Strategy for Development of Site-Specific Ubiquitin Antibodies. Front. Chem. 2020, 8, article 111.
